WPATH and Endocrine Society guidelines: Evaluation of evidence supporting the practice of pharmaceutical intervention in transgender adolescents
Avoiding Medical Reversal
This has turned into the longest piece I have written to date. I believe current medical practice in the United States runs the risk of doing great harm to some of our most vulnerable populations. The goal of this piece is to assess the evidence provided in guidelines by WPATH and the Endocrine Society.
The fundamental question is whether pharmaceutical interventions like puberty blockers and gender affirming hormones (GAH) are appropriate to give to adolescents with gender dysphoria.
In the United States, there are three relevant medical organizations for this question.
WPATH (World Professional Association for Transgender Health)
Endocrine Society
AAP (American Academy of Pediatrics)
These groups all support the practice of gender affirming care in youth. That is kind of a blanket term for things like social transition, puberty blockers, and GAH. Some claim that the support from these relevant organizations should be the end of the discussion. This is complicated by the fact that Sweden, Finland, the UK, and more recently Norway have all found the evidence behind gender affirming care to be lacking. The widening gap across the Atlantic in approach to gender dysphoria is summarized well in a recent article in the BMJ.
The Endocrine Society’s most recent clinical practice guideline in this space was updated in 2017. WPATH updated their guidelines in 2022. AAP has a short policy statement that largely defers to these two organizations, so we will focus on those guidelines.
Approach: In order to limit the scope of this review, we will directly assess the referenced studies with respect to the pharmacologic treatment of adolescents. Crucially, what evidence do we have today that these interventions improve mental health in gender dysphoric youth?
Note: The studies we will assess use a variety of terms that are interchangeable. For example, giving testosterone to trans boys is sometimes referred to as “cross-sex hormones” or “gender-affirming hormones”. The language used will reflect that of each paper that is being assessed.
Overview of Studies:
Endocrine Society: Two studies are referenced to support the benefit of pubertal suppression in gender dysphoric adolescents on mental health. This is actually a single cohort study with publications at two different time points. The quality of evidence behind this recommendation is rated as “low” by the Endocrine Society.
WPATH: The “Adolescents” chapter, updated in 2022, has a section titled “Research evidence of gender-affirming medical treatment for transgender adolescents.” Here the references come in a few waves:
5 longitudinal cohort studies, published from 1997-2014. These were largely conducted at one Dutch clinic. They looked at treatments including puberty suppression, affirming hormones, and surgeries.
7 more recent longitudinal studies published from 2015-2022. These look at the same interventions.
2 cross-sectional studies published in 2020 and 2021.
4 miscellaneous studies published from 2020-2022 (one chart review, one study of satisfaction with care, a healthcare data set study on healthcare utilization, and an internet convenience sample survey.
8 studies regarding the phenomenon of detransitioning. These last 8 studies we will not address, though it is clearly an important topic. The data to date just haven’t been collected in a systematic way.
WPATH does not report the quality of evidence behind the recommendations they give in this chapter. This is unfortunate, as it is standard practice that allows clinicians to understand whether the recommendation is based off things like randomized, controlled clinical trials or prospective cohort studies with no control arm.
Endocrine Society
The relevant clinical practice guideline, published in 2017, is linked here.
“Endocrine Treatment of Gender-Dysphoric/Gender-Incongruent Persons: An Endocrine Society* Clinical Practice Guideline”
If we go to the section on treatment of adolescents, we can look at specific “suggestions” they make and the evidence they cite to support those suggestions.
Use of pubertal suppression
2.1. We suggest that adolescents who meet diagnostic criteria for GD/gender incongruence, fulfill criteria for treatment (Table 5), and are requesting treatment should initially undergo treatment to suppress pubertal development. (2 |⊕⊕○○)
2.2. We suggest that clinicians begin pubertal hormone suppression after girls and boys first exhibit physical changes of puberty (Tanner stages G2/B2). (2 |⊕⊕○○)
Note: Those two plus signs in the circles indicate that the quality of the evidence behind the suggestion is “low”. One circle would mean very low, three means moderate, four means high. The use of the softer “suggest”, versus a stronger “recommend” is common when evidence is of low quality.
Claim: “Treating GD/gender-incongruent adolescents entering puberty with GnRH analogs has been shown to improve psychological functioning in several domains.”
The paper they cite for this claim is linked here.
“Puberty Suppression in Adolescents With Gender Identity Disorder: A Prospective Follow-Up Study”
This paper, published in 2011, followed 70 adolescents that were started on puberty blockers. They assessed psychological functioning and gender dysphoria two times. The first time was before they started puberty blockers, the second was shortly before the start of cross-sex hormone treatment. On average this was 2 years later.
Measures of psychological functioning:
The Child Behavior Checklist (CBCL) was filled out by parents
The Youth Self-Report (YSR) was filled out by adolescents
Beck Depression Inventory (BDI) filled out by adolescents
Trait Anger (TPI) and Anxiety (STAI) Scales of the State-Trait Personality Inventory was administered
Children’s Global Assessment Scale (CGAS) was performed by clinicians
Gender Dysphoria was measured using the Utrecht Gender Dysphoria Scale (UGS)
Results:
Psychological Functioning from T0 to T1: All measures improved except for anger and anxiety.
Gender Dysphoria: No change was observed.
My thoughts: There was a significant amount of missing data in these results. For CBCL and YSR they had data on 54 patients. For BDI, TPI, STAI, and CGAS they had data on 41 patients. Given the trial consisted of 70 participants, this represents a problem with interpreting the data. The authors mention that some assessment measures were added after the study had begun, making any patient that completed their initial measures prior to this unable to be assessed.
All adolescents were regularly seen by either psychologists or psychiatrists throughout the study. Most importantly for our purposes, there was no control group, no randomization.
It should be clear that this doesn’t allow us to conclude whether or to what degree pubertal suppression impacted these mental health outcomes.
Using GnRH Analogs
2.3. We recommend that, where indicated, GnRH analogues are used to suppress pubertal hormones. (1 |⊕⊕○○)
Claim: “Despite a decrease in depression and an improvement in general mental health functioning, GD/gender incongruence persisted through pubertal suppression, as previously reported. However, following sex hormone treatment and gender-reassignment surgery, GD/gender incongruence was resolved and psychological functioning steadily improved.”
This paper is from the same clinic as the first one we looked at, it is in fact the same cohort, later on.
“Young Adult Psychological Outcome After Puberty Suppression and Gender Reassignment”
55 of the original 70 participated in the study. The authors note this might have caused selection bias.
Reasons for non-participation of the 15 missing patients are listed below:
6 patients were not yet 1 year post-surgery
2 patients refused to participate
2 patients did not return questionnaires
3 patients were not medically eligible for surgery
1 patient dropped out of care
1 transfemale patient died after her vaginoplasty owing to a post surgical necrotizing fasciitis
We now have three time points. T0 was at patient intake, T1 was just prior to initiation of cross-sex hormones (CSH), and T2 was at least one year after gender reassignment surgery
Mean age for these timepoints were as follows: 13.6, 16.7, and 20.7 years of age for T0, T1, and T2, respectively.
Measures were similar to the previous study of this cohort. Additional measures were added at T2 only to further assess objective and subjective well-being
Results:
Psychological Functioning: Global functioning improved linearly over time. Depression increased directionally from T1 to T2. Over the entire length of the study (T0 to T2), there was no significant change in depression, anxiety, or anger.
Gender Dysphoria: Improved markedly from T1 to T2.
My Thoughts: We have a similar issue with missing data. The addition of further assessments led to 8 patients missing data at T0. Clinician error led to 10 patients missing data at T1. For CGAS, BDI, TPI, and STAI, data are reported for 32 patients. CBCL is reported for 40 patients. YSR is reported for 43 patients.
So we see in this study that gender dysphoria improves dramatically by one year after gender reassignment surgery. Global functioning in this cohort also improved with time, though depression, anxiety, and anger did not. It is tragic that a patient died from surgical complications, and I think untactful of the authors to include, “Whereas some studies show that poor surgical results are a determinant of postoperative psychopathology and of dissatisfaction and regret, all young adults in this study were generally satisfied with their physical appearance and none regretted treatment.”
Of course this study is limited by sample size, the short follow-up period after surgery, selection bias, and lack of any control group.
Two additional quotes from these studies provide context regarding the selectivity of these studies:
“At the Amsterdam gender identity clinic, adolescents are eligible for puberty suppression when they are diagnosed with GID, have shown persistent gender dysphoria since childhood, live in a supportive environment, and have no serious comorbid psychiatric disorders that may interfere with the diagnostic assessment. For example, it can be complicated to disentangle whether the gender dysphoria evolves from a general feeling of being just “different” or a whether a true “core” cross‐gender identity exists in adolescents who suffer from an autistic spectrum disorder.”
They go on to say:
“These individuals, of whom an even higher percentage than the general population were pursuing higher education, seem different from the transgender youth in community samples with high rates of mental health disorders, suicidality and self-harming behavior, and poor access to health services.”
The recommendations and claims mentioned previously from the Endocrine Society only cite these two studies of a single cohort of patients. They appropriately label the quality of the evidence as low and use the term “suggest” rather than “recommend”.
WPATH
The WPATH updated its guidelines in 2022 with SOC 8. “Standards of Care for the Health of Transgender and Gender Diverse People, Version 8”
We will go to the chapter on adolescents. The relevant section is titled “Research evidence of gender-affirming medical treatment for transgender adolescents”
WPATH acknowledges the dearth of data supporting medical treatments for gender-dysphoric youth. “..Therefore, a systematic review regarding outcomes of treatment in adolescents is not possible. A short narrative review is provided instead.”
Here they first mention five studies supporting the use of medical treatment for transgender adolescents. Two of these we covered in the Endocrine Society Guidelines. We will now look at the other three studies listed.
Link here, “Sex Reassignment of Adolescent Transsexuals: A Follow-up Study”
The study included the first 22 patients in the clinic who had undergone sex reassignment surgery at least one year previously.
2 patients refused to be interviewed, and one did not respond to the invitation. Final study included 14 FMs and 5 MFs.
They used interviews and questionnaires administered at pretest (mean age 17.5) and at follow-up (mean age 22.0). Mean elapsed time between the last operation and follow-up was two years.
Measures: They assessed a number of questionnaires and interviews to asses gender dysphoria and psychological functioning
9 of the 19 participants had started cross-sex hormones before the age of 18
Results: Gender dysphoria was dramatically reduced at follow-up. Psychological functioning showed limited effect.
The NVM personality inventory showed an increase in extroversion, with no significant change in negativism, somatization, shyness, or psychopathology
The NPV personality questionnaire showed a decrease in inadequacy and an increase in both dominance and self-esteem. No significant change in other measures (social inadequacy, rigidity, hostility, complacency).
My thoughts: This was a very small study (n=19) done in a time when medical intervention for patients with gender dysphoria was in its infancy. They found that in a small group of well-functioning patients, gender dysphoria had largely resolved as of follow-up ~2 years following surgery. Psychological benefits were modest, and of course there was no control group.
Our next study at the same clinic is linked here, “Adolescents With Gender Identity Disorder Who Were Accepted or Rejected For Sex Reassignment Surgery: A Prospective Follow-up Study”
This prospective study compared two groups.
The next 20 consecutive adolescents in the clinic who had undergone sex reassignment surgery (SRS). This is the treated (T) group.
21 applicants who either withdrew their request for SR, were rejected, or did not show up for appointments. This is the nontreated (NT) group.
Note: The authors acknowledge up front that these groups are not expected to be comparable. They say, “this study should be seen as an evaluation of two related but separate clinical decisions.”
The two clinical decisions they set out to evaluate are as follows:
“Whether it had been a correct decision to allow well-functioning adolescent transsexuals to proceed with the SR procedure after careful screening, given that they were between 16 and 18 years of age.”
“Whether the decision not to allow other adolescent applicants to proceed with the SR procedure before age 18 had been a justified one.”
Table 1 of the paper provides descriptive information about the NT group. There was an additional 6 patients who had delayed treatment, information on this group is also provided. Table 1 shows high rates of comorbid psychiatric conditions, severe family issues and sexual abuse.
Measures: They measured IQ, gender dysphoria, body dissatisfaction, and psychological functioning.
Demographics:
T group: mean age 16.6 at pretest, 21.0 at follow-up. Average of 1.3 years after surgery at follow-up. 10 of the T patients started hormone treatment between 16-18 years old.
NT group: Mean age 17.3 at pretest, 21.6 at follow-up.
Results:
Gender dysphoria: Gender dysphoria was reduced by ~75% in the treated group. Interestingly, it was reduced by ~33% in the non-treated group.
Body dissatisfaction: 3/3 measures improved in the T group. Only 1/3 improved (p<0.05) in the NT group.
Psychological functioning: They used two tests
NVM: None of the five psychological functioning traits changed in either group, though the baseline data give some insight into why many of the non-treated group were rejected. Their psychopathology score at baseline was over double that of the treated group.
SCL-90: T group improved with respect to hostility and depression. Anxiety and agoraphobia improved directionally but did not quite reach statistical significance. Psychoneuroticism, somatization, inadequacy, sensitivity, and sleep problems did not change. No improvement in any variable in the NT group.
My thoughts: This is a similar picture to the previous study. SRS in a small group of patients led to a major improvement in gender dysphoria at follow-up. The length of follow-up was short, making it difficult to draw conclusions about long-term outcomes. Psychological improvements were modest, which is not surprising, given how selective these studies are. The following quote from this paper demonstrates this well:
“Starting hormone treatment before adulthood should not be considered when many adverse factors operate simultaneously, despite the possibility that applicants may actually be transsexual. This is because it is more complicated to make an accurate diagnosis in problematic adolescents than in well-functioning adolescents, even for experienced multidisciplinary teams.”
The final of the five studies cited here we will skip, but it is linked here. I skip it because they studied adults, the mean age of participants that completed the study was 30.9 years at start of the study and 35.2 years at follow-up. I don’t know why it was cited in a section supporting evidence for intervention in adolescents.
Next the WPATH brings up seven more recent longitudinal studies that evaluate patients at different stages of treatment. We will go through an overview one by one.
Link here, “Longitudinal impact of gender-affirming endocrine intervention on the mental health and well-being of transgender youths: preliminary results”
This study was conducted in the United States, through Stony Brook University
From 2013-2018, 116 patients entered the study. 95 had never received endocrine intervention previously. Of these 95, 50 completed three waves of the questionnaires. Those 50 are analyzed in this report.
33 were Female to Male (FTM), 17 Male to Female (MTF). Baseline rate of depression and suicidality was high: 64% depressed in past year, about 10% reported suicidality. 90% of patients were receiving counseling, 34% were on a psychiatric medication.
FTM: By the third survey, 85% were on cross-sex hormones, 24% on puberty blocker, 12% on both.
MTF: By the third survey, 41% were on cross-sex hormones, 88% on puberty blocker, 41% on both.
Measures: In 6 months intervals they assessed depression (via CESD-R and PHQ-9) and quality of life (via QLES-Q-SF)
Results:
Overall: Both measures of depression improved. Quality of life directionally improved but did not reach statistical significance.
They ran a regression model to control for counseling and psychiatric medications, stratified by gender and endocrine intervention:
MTF participants on puberty suppression improved in one measure of depression (CESD-R)
FTM participants did not reach statistical significance for any measure.
My thoughts: This was a small study, with almost 50% drop out rate and no control group. The study lumps together puberty blockers and cross-sex hormones, and lacked the statistical power to assess these interventions in the regression analysis. As we have seen before, we are unable to assess causality.
Side note: This is the first study we have reviewed that was conducted in the United States. An important thing to keep in mind is that the first handful of studies we saw were all conducted within a rigorous framework of a multidisciplinary team that is highly selective. We can see in this paper that much of that scrupulousness is lost in this cohort:
“Most subjects were followed by mental health professionals. Those that were not were encouraged to see a mental health professional.”
Key word, encouraged. Not required.
Link here, “Well-being and suicidality among transgender youth after gender-affirming hormones.”
This study was conducted in the US, at Children’s Mercy Hospital
Participants were included if they had pretest and final assessment data points and received gender affirming hormones (GAH) for at least 3 months.
47 patients were eligible, 39 of whom received GAH only, 8 received puberty blockers followed by GAH.
They did two surveys assessing suicidality and general well-being
They found an increase in general well-being and a decrease in suicidality.
My thoughts: This was barely a study. They did two brief surveys of a small group of patients with no control arm. With respect to suicidality, they asked four questions that assess whether in the past few weeks they have experienced suicidal ideation (passive and/or active) or suicide attempt. Attempting to draw any conclusions from this study would be inappropriate.
Note: Once again I find it deeply troubling to read these papers from the US after having gone through the formative papers conducted in the Netherlands and elsewhere. We are so haphazard, as the following quote demonstrates: “To avoid unnecessary delays in medical care, our clinic does not require patients to be seen by one of our clinic’s mental health professionals if they have an established GD diagnosis and referral from a community mental health professional.”
Link here, “Psychosocial health in adolescents and young adults with gender dysphoria before and after gender-affirming medical interventions: a descriptive study from the Hamburg Gender Identity Service”
There were 434 patients in the study period, from 2013-2017.
At baseline, 124 chose not to participate, 40 had missing data (no informed consent or incomplete questionnaire sets)
270 patients completed the baseline questionnaires
66 were excluded due to being under 11y/o, having severe psychiatric problems, prior medical treatment, unfulfilled diagnostic criteria for GD, or diagnostic procedure still ongoing
204 adolescents eligible for follow-up
98 did not respond to follow-up invitation, 13 refused to participate, 14 had out-dated contact information, 4 dropped out (gave consent, but did not return questionnaire)
75 adolescents included in the follow-up analysis sample for a response rate of 37%
Note: I went through these details to highlight a common issue with many of these studies. Not only do they have no control arm, but we have no idea how the patients that refused to participate, dropped out, etc. may have had a different experience than the ones who agreed to continue with the study
64 were female (trans boys), 11 were male (trans girls)
Measures: Global functioning (via CGAS) and quality of life was assessed.
Results: “Adolescents who underwent puberty suppression or GA (hormonal and surgical) interventions showed better scores in some of the psychosocial health dimensions, although we did not test whether this difference was statistically significant.”
They didn’t have enough statistical power to test anything, so they were limited to a descriptive analysis. The authors point out that this kind of analysis cannot be compared to other samples.
My thoughts: This was a small study with a huge level of attrition. They had a pseudo-control group, given something like 25% of the cohort didn’t receive medical treatment. But they also note this doesn’t allow ready comparison because patients with poorer mental health are less likely to be treated. This study is unable to tell us anything.
Link here, “Short-term outcomes of pubertal suppression in a selected cohort of 12 to 15 year old young people with persistent gender dysphoria in the UK”
They gave 44 patients (12-15y/o) GnRHa in an uncontrolled prospective observational study of adolescents with persistent and severe GD.
This was a GIDS clinic study (UK)
Treatment duration ranged from 1-5 years, 3.75mg triptorelin intramuscular injection every 28 days. Regular attendance for psychological support and therapy throughout the study was provided.
Only one patient decided to stop GnRHa and not start cross-sex hormones, “due to uncertainty and some concerns about side-effects of cross-sex hormones”. The other 43 went on to take cross-sex hormones.
Results:
Bone Mineral Density (BMD): Lumbar and hip bone mineral density age-adjusted z-scores were in normal range at baseline, they decreased at 12 and again at 24 months, and stayed flat at 36 months. Height adjusted z-scores for lumbar and hip BMD also followed the same pattern.
Psychological Outcomes: There were no significant changes in any of the psychological parameters. No change in CBCL self-harm index nor self-report YSR self-harm index scores.
Participant experience, satisfaction:
6-15 months: 46% reported only positive changes, 37% reported both positive and negative changes, 12% reported only negative changes, 5% reported no change
15-24 months: 55% reported only positive changes, 17% reported both positive and negative changes, 17% only negative, 10% no change
Side effects:
Mild headaches or hot flushes were reported in 25% at 0-6 months, 23% at 7-12 months, and 22% at 13-24 months
Moderate or severe headaches and hot flushes were reported in 5% at 0-6 months, 9% at 7-12 months, and 3% at 13-24 months
Mild fatigue was reported in 5-8% of patients
My Thoughts: Small study, no control group. No evidence of change in psychological function or self-harm. Pubertal suppression led to decreased BMD z-score.
Note: In the discussion section of the paper they bring up two possible explanations for the fact that pubertal suppression led to no change in psychological wellbeing or self-harm. The authors say it could be that this intervention truly has a neutral effect, or it’s possible that the intervention was beneficial and it masked what otherwise would have been a worsening of psychological functioning.
While I agree each of these are possible, they omitted the third obvious possibility. It’s possible that therapy/counseling without the pubertal suppression would have led to improved psychological functioning and reduced measures of self-harm, and the medication exerted a negative effect.
This is of course speculative, but such are the questions left unanswered by the lack of any control group.
Link here, “Psychological Support, Puberty Suppression, and Psychosocial Functioning in Adolescents with Gender Dysphoria”
This study was conducted at GIDS in London.
From 2010-2014, 436 adolescents were referred to GIDS. 201 adolescents completed the diagnostic procedure and were invited to take part in follow-up.
Eligible adolescents were divided into two groups: immediately eligible and delayed eligible adolescents
Delayed eligible adolescents were those which clinicians needed more time to make the decision to start pubertal suppression due to possible comorbid psychiatric problems and/or psychological difficulties. These patients were referred to local mental health services. None of the delayed eligible individuals received pubertal suppression at the time of this study.
Measures included GD-related discomfort (via Utrecht GD Scale, or UGDS) and global psychosocial functioning (via Children’s Global Assessment Scale, or CGAS)
These were measured at baseline, 6 months, 12 months, and 18 months
They had a high drop-out rate after the 6 month assessment. Below are the sample sizes by assessment time (n= baseline, 6 months, 12 months, 18 months)
Immediately eligible: n= 101, 101, 60, 35
Delayed eligible: n=100, 100, 61, 36
Results:
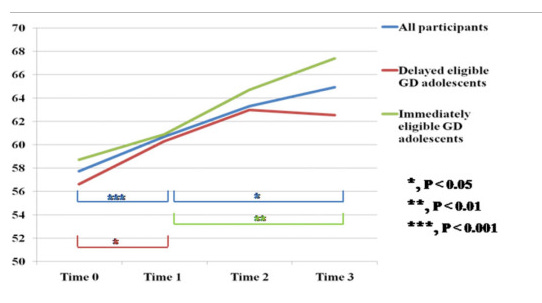
Here we can see the trend in global psychosocial functioning.
Conclusion by the authors: “In conclusion, this study confirms the effectiveness of puberty suppression for GD adolescents.”
My thoughts: No it doesn’t. This is really irresponsible. You don’t get to make a claim in the scientific literature without strong evidence. Language in medical literature can be dry, but saying “this study confirms” is an emphatic claim! Let’s limit this analysis to the crucial flaws in this study.
They didn’t have a comparable control arm. The group that only received psychological/psychiatric intervention were deemed by their clinician at GIDS to not be ready for puberty blockers. It wasn’t randomized, and the graph clearly shows the delayed group started at a lower CGAS score.
They had an incredibly high dropout rate! This goes bizarrely unmentioned in the “limitations” section of the discussion. They cite a small sample size, but no mention of the fact that by 12 months each arm had lost ~40% of the participants, and by 18 months they had lost ~ 65%. Why did they lose so many adolescents? The only follow-up point we can have confidence in being accurate is the 6 month follow-up, at which point 100% of participants were accounted for.
If we look at 6 months on the graph above, delayed participants nearly matched their peers on puberty blockers with respect to global psychosocial functioning.
Link here, “Body Dissatisfaction and Mental Health Outcomes of Youth on Gender-Affirming Hormone Therapy”
This study had three goals:
To assess baseline differences in body dissatisfaction, depression, and anxiety by gender, age, and pubertal stage.
To assess for changes in these measures over the first year receiving gender-affirming hormone therapy.
Examine potential differences in change over time by demographic and treatment characteristics
148 participants, ages 9-18, mean age 14.9 were receiving gender-affirming hormone therapy in Dallas, Texas. 25 were receiving pubertal suppression only, 123 received feminizing or masculinizing hormone therapy.
They assessed participants at baseline and roughly one year later.
209 participants were eligible, 148 provided follow up data. Reasons for the missing participants listed below:
3 did not consent
4 had no follow-up after initial assessment
11 were prepubertal during follow-up period
8 were already being treated at baseline
4 did not have parental consent
11 transferred care to another provider
2 chose to stop pubertal suppression
1 lost to follow-up
17 missed follow-up
Results: Participants had decreases in body dissatisfaction, self-reported depressive symptoms, and total anxiety symptoms during follow-up. No change in clinician report of depressive symptoms was observed. The effect size was:
Large with respect to improvement in body dissatisfaction
Small to moderate for self-report depression
Small for total anxiety scores
Suicidal ideation, suicide attempt, and nonsuicidal self-injury (NSSI):
Lifetime rate of passive suicidal ideation (SI) was 81%, Suicide attempt was 15%, NSSI was 52%.
Passive suicidal ideation: 25% of patients reported passive SI in the 1-3 months before initial assessment. 38% had passive SI during the follow-up period.
Suicide attempt: 2% of patients reported a suicide attempt in the 1-3 months period before initial assessment. 5% had a suicide attempt during follow-up period
NSSI: 10% of patients reported NSSI in the 1-3 months before initial assessment, 17% of patients during follow-up period
Conclusions from the authors: “The current study is the largest longitudinal study of youth receiving gender-affirming hormone therapy to date and documents important improvements in body dissatisfaction over the first year of treatment.”
My thoughts: I’m much happier with these authors than those of the previous study. They did document improvement in body dissatisfaction over the first year of treatment. Can confirm. The trouble of course is we are no closer to learning whether these treatments are appropriate. No control group, minimal to no improvement in mental health, and by no means did they demonstrate that these interventions will reduce suicidal thoughts, suicide attempts, or self-injurious behavior.
Link here, “Mental Health Outcomes in Transgender and Nonbinary Youths Receiving Gender-Affirming Care”
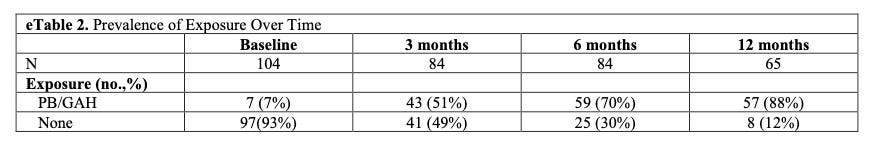
This was a prospective cohort study of 104 patients ages 13-20 receiving care at Seattle Children’s Gender Clinic
All patients who completed phone intake and in-person appointments from August 2017- June 2018 were recruited.
Baseline surveys were completed within 24 hours of their first appointment, follow-up surveys were sent at 3, 6, and 12 months.
Measures: Depression (via PHQ-9), anxiety (via GAD-7), and suicidality (via question 9 on PHQ-9
169 patients were screened, 161 were invited to participate. 9 declined, 39 did not complete consent or assent or did not complete the baseline survey. 9 patients were younger than 13 and were excluded due to differences in screening protocol.
104 patients ages 13-20 participated. Follow-up surveys were completed by 84, 84, and 65 patients at months 3, 6, and 12, respectively.
Results:
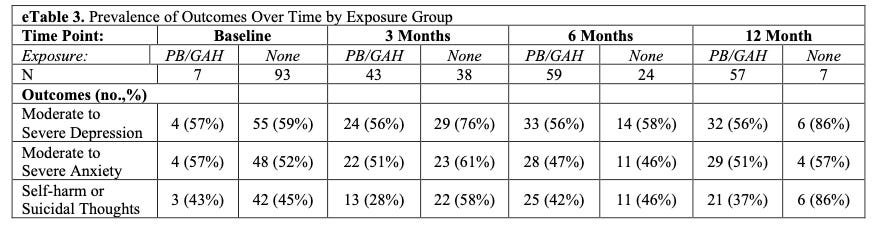
For full data set analysis: No statistically significant trends throughout the study. The tables below give the most direct information, allowing interpretation.
“There were a small number of youth who did not complete the PHQ-9 or GAD-7 on each survey: 4 youth at baseline, 3 youth at 3 months, and 1 youth at 6 and 12 months.”
“…we observed that youths who had initiated PBs or GAHs had 60% lower odds of moderate to severe depression (aOR, 0.40; 95% CI, 0.17-0.95) and 73% lower odds of self-harm or suicidal thoughts (aOR, 0.27; 95% CI, 0.11-0.65) compared with youths who had not yet initiated PBs or GAHs.”
For patients that did not receive PBs or GAHs, they had statistically significant increase in moderate or severe depression at 3 months, which lost significance at 6 and 12 months. They found a similar increase in self-harm or suicidal thoughts at 6 months, which further increased directionally but lost statistical significance at 12 months.
Conclusions from the authors: “Our study results suggest that risks of depression and suicidality may be mitigated with receipt of gender-affirming medications in the context of a multidisciplinary care clinic over the relatively short time frame of 1 year.”
My thoughts: This paper has some odd features.
First let’s start with the usual limitation, no control group again. This study essentially used the patients as their own controls, but pretended they had two separate groups. At the start, 93% of patients were not on hormones, only 7% were already taking them. By the end, 88% were on hormones, only 12% were not. You don’t get to pretend that patients without exposure to hormones represent a control arm when that arm starts with 97/104 patients and ends with 8/65 patients. This is a fatal flaw for any comparison of patients that did vs. did not receive hormones in this study.
Attrition, again. They started with 104 patients at baseline, dropped to 84 at 3 months, stayed there at 6 months, and dropped to 65 at 12 months. So they lost almost 40% of the study population. What happened to those kids? We have no idea. This is a massive flaw that we have seen before.
Outcome measures: They did this bizarre thing where the outcome was dichotomized. They used a cutoff of 10 or more for both the PHQ-9 and GAD-7. We don’t get to see the data on how much these scores actually changed. A patient going from a 10 to a 9 and a different patient going from 10 to 1 are scored the exact same way. This is unhelpful.
Who was measuring: In a previous study we saw that self-reported depression scores improved modestly, but when these variables were assessed by a clinician there was no difference. In this case they just sent out some surveys, they did not have a clinician assess these patients.
What did they find? I don’t think there is a baby in this bathwater, but just for fun: The only halfway reasonable way to try and make use of data like this is to look at the complete data set, and probably to remove the 12 month time point given the huge drop-off in follow-up. With or without that time point, there was no overall trend in any of the outcomes.
CROSS SECTIONAL STUDIES
Next we are given two cross-sectional studies.
Link here, “Psychological Functioning in Transgender Adolescents Before and After Gender-Affirmative Care Compared With Cisgender General Population Peers”
This was a cross-sectional study of 272 adolescents referred to a specialized gender identity clinic, all of whom had not received any affirmative medical treatment.
They were compared to two groups:
178 transgender adolescents receiving affirmative care via pubertal suppression
651 Dutch high school cisgender adolescents
This was done in Amsterdam, patients were referred between 2012 and 2015. The cisgender control group was recruited in 2015.
More details on study arms:
272 adolescents who just started the assessment process (mean age 14.5)
178 adolescents who were on puberty suppression and about to start GAH (mean age 16.8)
651 cisgender adolescents from general population (mean age 15.4)
Results: Adolescents that had just been referred to a specialized gender identity clinic had significantly higher scores than either comparison group with respect to internalizing, peer relations, and suicidality. Transgender adolescents using puberty suppression scored similarly to cisgender peers with respect to internalizing and self-harm/suicidality, but scored higher on peer relation problems.
My Thoughts: Cross-sectional data gives very little information on effects from treatments, for obvious reasons.
The authors have a nice section in which they emphasize that the approach in the Netherlands to treatment with puberty blockers is far more rigorous than other countries, an important point for anyone that would attempt to extrapolate their data to say, the United States. It is quite impressive that these adolescents are scoring similarly to cisgender peers for most measures.
We have some common issues, like the reliance on self-report and not clinical evaluation. More importantly there is no coherent way to draw causative conclusions from a cross-sectional study like this.
We have no ability to assume that adolescents who were just referred to the clinic are comparable to the older adolescents on pubertal suppression. The authors are appropriately measured in their acknowledgment of this: “The present study can, therefore, not provide evidence about the direct benefits of puberty suppression over time and long-term mental health outcomes.”
Link here, “Testosterone treatment, internalizing symptoms, and body image dissatisfaction in transgender boys”
They found adolescent transgender boys (natal females), 19 of whom were taking Testosterone (T), and 23 who were not (UT)
47 patients were included. Four participants did not complete the protocol, one was excluded due to a chromosomal condition.
None of the participants had previously received puberty blockers.
Differences between patients receiving T and not (UT)
T group was older, average age 17.03, versus 15.75 in UT group (statistically significant difference)
53% of the T group was on anxiolytics/anti-depressants, versus 78% of UT group
T group scored lower on SRS, a measure associated with Autism Spectrum Disorder, with an average of 65.8 versus 76.2 in the UT group.
They did some self-report measures for anxiety, depression, body image dissatisfaction, and self-harm/suicidality.
The investigators did fMRI scans, looking at activation of the amygdala and its connectivity to the PFC. We can just ignore this portion of the study because it's just so far from being relevant to our central question in this review.
Results: The T group had significantly lower levels of generalized anxiety, social anxiety, and depression than the UT group. Though not significant, directionally the T group had a lower rate of suicidality. The T group also had significantly lower levels of body dissatisfaction than the UT group.
Authors Claim: “A primary finding of this study is that symptoms of anxiety, depression, and suicidality were lower in testosterone-treated transgender adolescents than in a comparable group of transgender adolescents not receiving GAH.”
My Thoughts: The above claim would have been better if they didn't include suicidality. It’s sloppy to lump something that wasn’t statistically significant in with the measures that were. Kind of a bad look to say the “primary finding” of your study includes something that your study failed to show a clear difference in.
The comparison group is clearly different than the T group, as Table 1 of the study shows.
This is cross-sectional, not longitudinal, so were are unable to infer any kind of causation.
Confounders are numerous (eg, therapy, comorbid psychiatric conditions, etc.)
Sample size is small
We have four final studies cited by WPATH that we will assess.
Link here, “Adolescent development and psychosocial functioning after starting cross-sex hormones for gender dysphoria”
This was a retrospective chart review of 52 adolescents in Finland.
All were assessed before the age of 18, all were started on hormonal gender reassignment.
Between 2011 and 2017, 57 adolescents were offered hormonal sex reassignment. One declined treatment, two withdrew and two had started but not spent enough time (one year) on treatment to be included.
52 patients were included, mean age of 18.1 at diagnosis
Measures included indicators of adolescent development (eg, living arrangements, romantic involvement, need for psychiatric treatment, etc.)
Results: No statistically significant improvements in functioning were observed in the group as a whole.
Conclusion from the authors:
“Among adolescents diagnosed with transsexualism, difficulties in adolescent development and functioning in life domains appropriate to late adolescence do not disappear with cross-sex hormone treatment. Cross-sex hormone treatment may alleviate depression and anxiety but does not have a positive impact on psychiatric comorbidities at large. Even deterioration as regards psychiatric treatment needs and functioning occurs during the first year of cross-sex hormone treatment. Not all psychiatric and psychosocial problems in adolescents displaying gender dysphoria are secondary to gender identity issues and will not be relieved by medical gender reassignment. An adolescent’s gender identity concerns must not become a reason for failure to address all her/his other relevant problems in the usual way.”
My thoughts: I quoted the conclusion at length because I thought it was well put. While there was no change in the need for psychiatric treatment, there was an encouraging decrease in the reason listed for that treatment being depression, anxiety, or suicidality/self-harm. Interpreting the significance of that is difficult given we aren’t provided with an explanation for what was driving the need for psychiatric treatment, nor what that treatment entailed (talk therapy, medications, etc). It goes without saying there was no control group.
Also, must be nice… “Due to excellent social security benefits, moving to live independently does not necessitate regular income from employment and is therefore not a proxy for good functioning in other domains of life.”
Link here, “Individual Treatment Progress Predicts Satisfaction with Transition-Related Care for Youth with Gender Dysphoria: A Prospective Clinical Cohort Study”
434 children and adolescents were originally included, this was done at the Hamburg GIS
Drop-out at baseline: 124 did not participate, 40 were missing data.
Exclusion criteria: 38 children were under 11 years old, 8 adolescents had severe psychiatric problems, 2 had prior medical treatment, 6 didn’t meet diagnostic criteria for GD, 12 diagnosis was not yet established at study start
204 adolescents eligible for follow-up, 98 did not respond to invitation, 13 refused to participate, 14 had outdated contact information, 4 dropped out (consent given, no questionnaires returned)
75 adolescents were in the final sample
Mean age 17.4 years at the time of follow-up.
54 participants had received transition-related medical interventions (TRMI) at follow-up. 21 had not received any such interventions.
At follow-up, 11 youth had received GnRHa, 32 adolescents had received GAH, and 11 had received both.
Measures were focused on satisfaction with treatment, assessed via survey (ZUF-8)
Results: “Based on the scores on the ZUF-8, 75% of all participants indicated that they were overall satisfied with the TRC received at the Hamburg GIS…”
My Thoughts: This study isn’t helpful to assess appropriateness of these interventions. They had a 37% response rate. It was a survey that showed people who continued to seek treatment and progress down the pathway of care generally were satisfied with that care. No control group. We’re getting pretty thin here with these studies.
Link here, “Mental Healthcare Utilization of Transgender Youth Before and After Affirming Treatment”
Retrospective cohort study using United States military healthcare data from 2010-2018.
3,754 transgender and gender-diverse (TGD) adolescents and 6,603 cisgender siblings were included.
Baseline:
“TGD adolescents were more likely to have a mental health diagnosis (OR 5.45, 95% CI [4.77–6.24]), use more mental healthcare services (IRR 2.22; 95% CI [2.00–2.46]), and be prescribed more psychotropic medications (IRR = 2.57; 95% CI [2.36–2.80]) compared to siblings.”
Results:
“Among 963 TGD youth (Mage: 18.2) using gender-affirming pharmaceuticals, mental healthcare did not significantly change (IRR = 1.09, 95% CI [0.95–1.25]) and psychotropic medications increased (IRR = 1.67, 95% CI [1.46–1.91]) following gender-affirming pharmaceutical initiation; older age was associated with decreased care and prescriptions.”
My thoughts: This was a really short one, the cliff notes give us most of the information. The authors observed a decrease in the median number of mental healthcare visits after gender-affirming pharmaceuticals. This wasn’t statistically significant in the adjusted regression analysis. The increase in psychotropic medications following treatment with gender-affirming medications is concerning, but like the other studies it has to be taken in context.
The main strength is definitely the sample size, as well as the objective data on diagnoses, mental health visits, psychotropic medications, sibling controls, etc. It doesn’t suffer the flaws of self-report, sample size of 40, etc.
Some of the limitations include:
Reliance on ICD codes, which is a binary measure that gives limited information.
It’s not clear that the lack of a decrease in mental health visits overall means anything clinically, though the increase in psychotropic medications is a more clearly undesirable outcome.
The unique population being military families limits generalizability.
Link here, “Access to gender-affirming hormones during adolescence and mental health outcomes among transgender adults”
Another cross-sectional study. They looked at the 2015 U.S. Transgender Survey, which includes 27,715 transgender adults
They looked for association between access to GAH during early adolescence (14-15), late adolescence (16-17), or adulthood and adult mental health outcomes.
They split survey respondents into 4 groups
Desired GAH, never accessed
Desired GAH, accessed during early adolescence
Desired GAH, accessed during late adolescence
Desired GAH, accessed in adulthood
Results:
GAH during early adolescence: They report a decrease in past-month severe psychological distress and past-year suicidal ideation (SI) when compared to desiring GAH but never accessing them.
No difference in SI with plan, suicide attempt, binge drinking, lifetime illicit drug use.
GAH during late adolescence: Same pattern as above.
GAH during adulthood: Same pattern except they had a higher rate of past-month binge drinking and lifetime illicit drug use.
They went on to do some post-hoc analyses that weren’t included in the statistical plan, but let’s take a look anyways.
They conflated the two GAH in adolescence groups, compared them to GAH in adulthood, and found lower odds of psychological distress, SI, binge drinking, and lifetime illicit drug use in the adolescence group.
My thoughts: This study was pretty tiresome, it reminds me of reading literature in nutrition. Survey results are quite limited even in the clinical research setting we have seen so many times in this review. Now we’re supposed to make inferences based off a single survey sent out to an internet convenience sample? Idk, it just feels like a stretch, not worth the time.
The next part of the WPATH chapter on adolescents considers the topic of regret and detransition. We will not assess these studies, as there is little certainty on this topic. It largely has just not been studied.
This brings us to the end of the section on evidence for gender-affirming medical treatment for transgender adolescents. The WPATH authors finish with this:
“Taken as a whole, the data show early medical intervention—as part of broader combined assessment and treatment approaches focused on gender dysphoria and general well-being—can be effective and helpful for many transgender adolescents seeking these treatments.”
They go on to give a series of specific recommendations. None of these recommendations indicate the quality of the evidence behind them. We have reviewed that quality in this document. To summarize:
18 studies total. There were zero randomized, controlled trials. There were zero randomized trials. I also think we have seen that when a control arm was present, it was never readily comparable to the treatment arm. The authors of these studies were consistent in acknowledging this.
One big issue this leaves us with is we simply don’t know whether pharmaceutical intervention is appropriate in adolescents. Some argue it would be unethical to conduct RCTs for these interventions because they believe they are effective. This doesn’t make sense, as we don’t know if they are effective. We do know that almost every individual started on puberty blockers will go on to take cross-sex hormones. Referring to puberty blockers as fully reversible is at best highly misleading, even if we ignore the lack of long-term data on their side effects.
I think some of the Dutch studies do show that cross-sex hormones can alleviate gender dysphoria specifically. They overall showed very little impact on mental health. At the risk of oversimplifying, the approach they used in some ways treated hormonal intervention as a “treatment of exclusion”. We have seen them state multiple times that the mental health of the patients they put on these treatments are roughly comparable to the general population. Educational attainment was higher in at least one study. They did not have disproportionate rates of suicidality, psychiatric comorbidities, etc.
I fear that measured approach is undergoing “McDonaldization” in the US. Of course the cultural furor around this topic does not help. Combine our lack of dispassionate scrutiny with the massive profitability of medicalizing these adolescents and I’m afraid you get a perfect set up for a medical reversal.
Very few people would agree that a minor is capable of consenting to treatment that can lead to infertility. The common counterpoint to this claim is to state that these individuals are at high risk of suicide, and these interventions reduce that risk. That reduction has simply not been demonstrated. Not a single study we reviewed was able to determine the effect of puberty blockers or cross-sex hormones on the risk of suicide. We have zero studies that assess therapy vs. therapy + hormonal intervention in comparable groups. We have failed to do the appropriate studies to determine what benefit or harm is derived from these treatments.
Books have and will be written on the complexities of this topic. My goal here was to go directly to the two organizations most deferred to in the Unites States regarding this topic. We assessed the studies that were cited as evidence supporting the use of pharmacological intervention in transgender adolescents in each of their respective clinical guidelines. I personally have found them lacking. Our most vulnerable youth will reap what our medical institutions sow.


This is an incredible breakdown. I am a med student as well and admire your work. It's very difficult in our current climate to question pro-vaccine (or pro-treatment) data. As a researcher I was taught to question everything, but in medicine that is not tolerated. Like you, I have studied GD treatment and WPATH in great depth and you have brought up the many concerns that most are either not intelligent / thoughtful enough, too lazy, or too imbibed in group think to raise themselves.
I just read this after reading your analysis of data re RSV vaccine this AM. I'm a pediatrician and very concerned about the poor evidence basis for this very consequential treatment in young people. I haven't seen a summary of all of this data in such a clearly written format anywhere else, and really appreciate it. It doesn't look like you got much response when you posted this, which must have been a bit discouraging. I hope you'll keep it up anyway! Thanks again!